
All matter in the universe is made up of basic building blocks. These tiny components, which form everything around us, possess unique characteristics that define their behavior and interactions. By examining these basic units, we gain insight into the makeup of everything from the smallest objects to the most complex systems.
Each unit contains several distinct components, each with a specific role. Together, they work in harmony, contributing to the unit’s overall stability and function. Knowing how these individual elements are arranged provides a clearer picture of how matter behaves on both a microscopic and macroscopic level.
In the following section, you will be guided through a visual representation, helping to identify and understand the role of each element within these fundamental units. As you explore this structure, consider how each component plays a crucial role in the larger framework of matter.
Understanding Atom Structure and Components
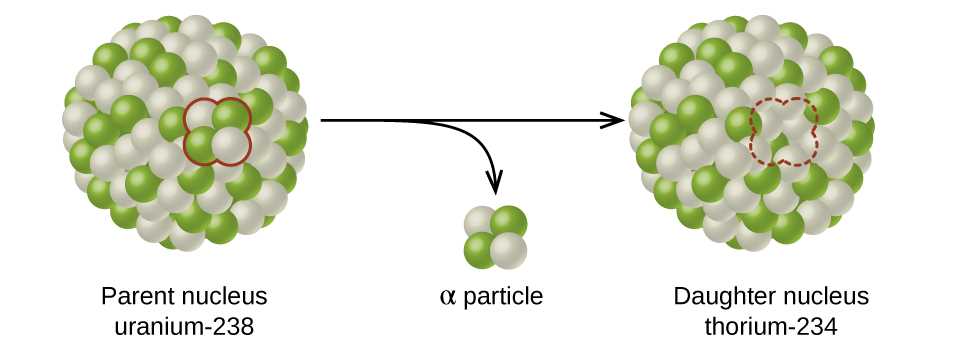
In this section, we will explore the core aspects of matter’s fundamental building blocks. These microscopic entities form everything around us, playing a crucial role in both chemical reactions and physical properties. Gaining insight into their internal makeup helps explain how substances behave and interact.
At the heart of this exploration lies an intricate system composed of different elements, each fulfilling a distinct role in maintaining stability and function. By understanding their arrangement and interactions, we unlock the key to countless scientific phenomena and technological advancements.
This structured breakdown of components reveals how energy, mass, and charge are distributed within this entity, explaining how they govern forces that hold everything together. A deeper appreciation for these interactions can enhance knowledge in fields such as chemistry, physics, and engineering.
Identify the Key Atomic Particles
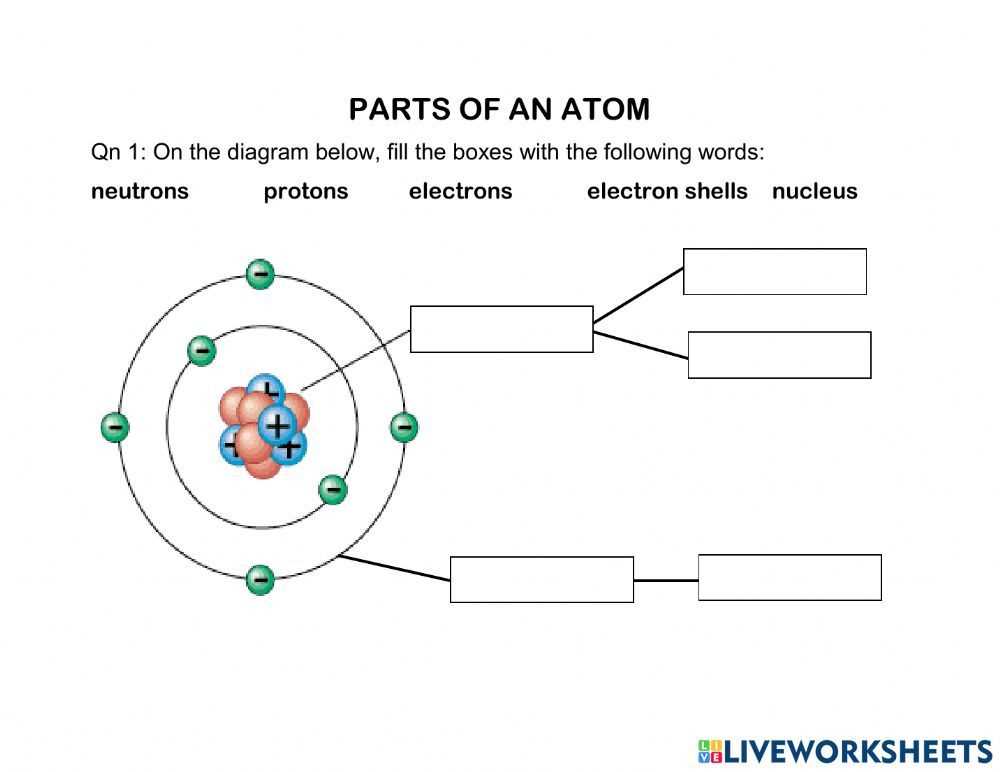
Every element is made up of tiny constituents that interact to define its fundamental nature. Understanding these building blocks reveals the basic structure and how these units behave under different conditions. Each component plays a critical role in determining properties like mass and charge, influencing everything from chemical reactions to physical phenomena.
Main Subatomic Units
At the core of this structure, several distinct entities coexist. One type, known for its positive charge, resides centrally, while another neutral entity shares this space. A third type, negatively charged, orbits around, moving rapidly in specific paths. Together, these units maintain a delicate balance that governs stability.
Interaction Between These Units

The interplay between positively charged and negatively charged particles results in an attractive force that keeps the structure cohesive. This interaction is key to understanding how different elements combine and interact, shaping the world on both microscopic and macroscopic levels.
Electrons and Their Orbital Behavior

Electrons are fundamental to how matter interacts and forms bonds. These subatomic particles are dynamic, constantly moving in complex paths that define their relationship with other particles. Understanding their movement helps explain chemical reactions and energy transfers.
- Electrons travel in specific regions known as orbitals, which are based on energy levels and quantum mechanics.
- Each orbital can hold a limited number of electrons, influencing how elements form compounds.
- Orbitals come in different shapes, including spherical and dumbbell-like configurations, which affect interactions with nearby particles.
The behavior of electrons in these orbitals determines much of what we observe in chemistry, from the structure of molecules to reactions between substances. Their ability to absorb and release energy also plays a crucial role in processes like electricity conduction and light emission.
Protons: The Nucleus’s Positive Charge

Protons are fundamental building blocks, residing in the core of every element. Their presence defines the core’s charge and plays a crucial role in determining the identity of an element. Together with other subatomic entities, protons ensure the stability of the core and influence the overall behavior of matter.
Here’s a closer look at the key characteristics of protons:
- Positive Charge: Each proton carries a positive charge, crucial for balancing negative particles within a structure.
- Mass: While tiny, protons have significant mass compared to some other particles, contributing to the overall weight of matter.
- Interaction with Neutrons: Protons work alongside neutral particles to maintain stability in the core, preventing decay.
- Defining Element Identity: The number of protons is unique for every element and determines its chemical properties.
In summary, protons are vital to the structure and behavior of all matter, their charge influencing interactions and defining the nature of each element.
Neutrons: Neutral Particles in the Core

Neutrons are essential elements found at the center of matter. Their main role is to balance forces within the core, ensuring stability in various structures. While not possessing any charge, they still significantly influence interactions.
Being neutral means that these particles neither attract nor repel other charged components. Instead, they contribute to the cohesion and overall structure. Without neutrons, many entities would be unstable, leading to transformations in their behavior.
Understanding the role of neutrons helps to explain how different materials and elements hold together, highlighting their importance in maintaining balance in the core of everything.
Electron Clouds and Energy Levels

Understanding how electrons are distributed around a nucleus is essential for grasping the behavior of matter. Electrons do not follow fixed paths but instead occupy regions that are more like probability zones, known as electron clouds. These regions help explain the movement and organization of electrons in different energetic states.
The concept of energy levels plays a crucial role in this arrangement. Electrons are grouped based on how much energy they possess, and their positions influence how they interact with other entities. The higher the energy level, the further the electron is likely to be from the nucleus, giving a better picture of how chemical reactions and bonding occur.
The Role of Atomic Number
The atomic number plays a crucial role in defining the identity of an element. It represents a fundamental property that distinguishes one element from another and determines various characteristics of matter. Understanding this number helps to explain chemical behavior and how different elements interact with each other.
- Determines the type of element
- Defines chemical properties
- Influences reactions and bonding patterns
By grasping how the atomic number affects the structure of matter, scientists can predict the behavior of elements in different environments, aiding in advancements across chemistry, physics, and material sciences.
Mass Number and Isotopic Variations
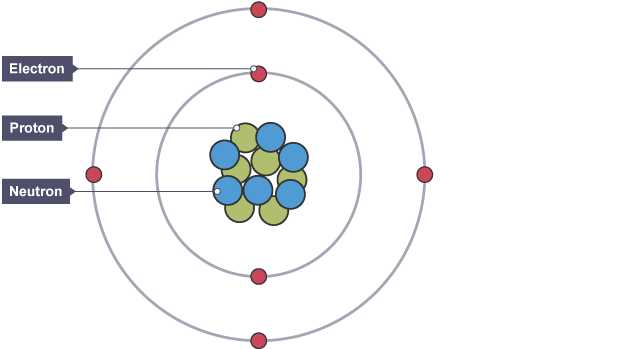
Understanding the concept of mass number helps to explain why not all examples of a certain chemical element are identical. The idea revolves around variations in the total number of certain subatomic components, leading to different forms of the same element. These forms are often studied to gain insights into stability, behavior, and usage in scientific applications.
Below are key aspects of mass number and isotopic variations:
- Mass number refers to a specific feature related to subatomic components and can vary between different examples of an element.
- Isotopic forms result from changes in specific particles within these structures, even though chemical properties remain largely consistent.
- Differences in these forms may lead to variations in behavior, including in nuclear reactions or stability over time.
These factors contribute to the diversity seen in nature and scientific study, where isotopic forms play crucial roles in fields such as chemistry, medicine, and environmental science.
Diagram Labeling for Atomic Clarity
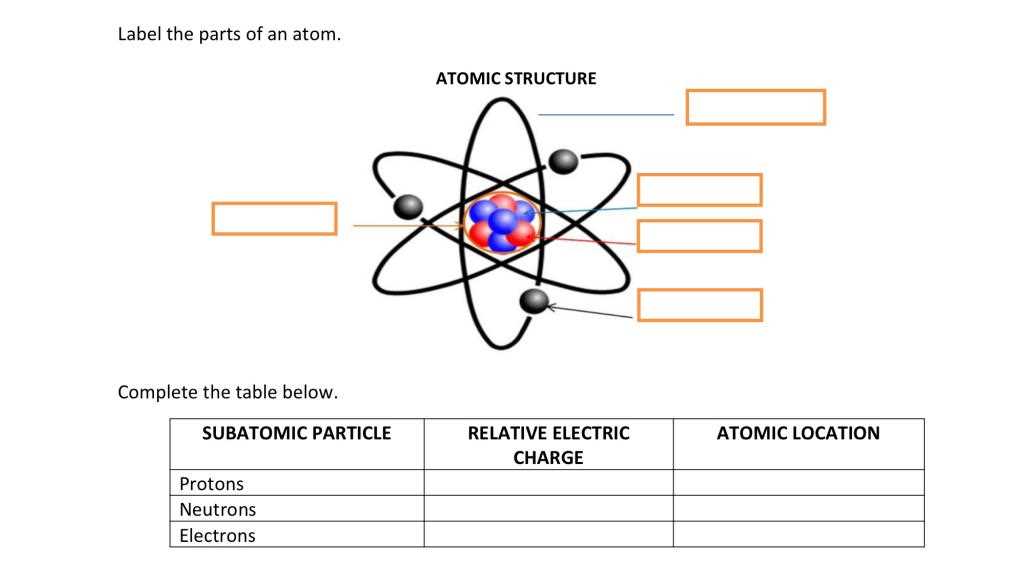
Understanding atomic structures can be simplified by focusing on individual components and their interactions. This approach enables a clearer grasp of how subatomic entities function together to form the foundation of matter. Rather than getting lost in technicalities, a well-organized representation will break down the complex structure into easily recognizable segments.
- Focus on recognizing core elements of the atomic structure.
- Understand how these components interact to maintain balance.
- Visual aids help reinforce comprehension of the relationships between the atomic entities.
- Organized presentation facilitates the learning process.
By identifying and distinguishing these core entities, the abstract nature of atomic theory becomes more approachable. A clear visual representation transforms complexity into manageable insights.